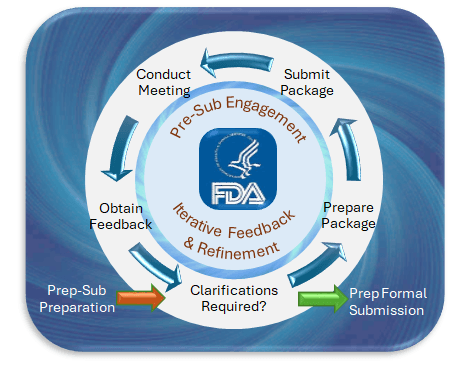
On Dec. 4, 2024, the U.S. Food and Drug Administration (FDA) issued a final guidance on “Marketing Submission Recommendations for a Predetermined Change Control Plan (PCCP) for Artificial Intelligence-Enabled Device Software Functions.” This guidance provides a framework for manufacturers to proactively plan for modifications to AI-enabled medical devices, ensuring that these updates maintain device safety and effectiveness without necessitating new marketing submissions for each change.
PCCP – What is it?
A Predetermined Change Control Plan is a comprehensive document that outlines anticipated modifications to a device’s software and the methods for implementing and validating these changes. For AI-enabled devices, which often require iterative updates to improve performance or adapt to new data, a PCCP allows manufacturers to:
Define Specific Modifications: Clearly describe the types of changes anticipated, such as algorithm updates or performance enhancements.
Establish Validation Protocols: Detail the procedures for assessing the impact of these modifications on device performance and safety.
Set Implementation Strategies: Outline how and when the modifications will be integrated into the device’s software.
By including a PCCP in the initial marketing submission, manufacturers can implement specified changes without submitting a new application for each modification, provided these changes fall within the scope of the approved PCCP.
Impact on the Medical Device Industry:
The FDA’s guidance on PCCPs will significantly influence the medical device sector, particularly in the realm of AI-enabled technologies:
Accelerated Innovation: Manufacturers can rapidly update their devices to incorporate the latest advancements in AI, ensuring that patients and healthcare providers have access to cutting-edge solutions.
Regulatory Efficiency: The ability to implement pre-approved changes without additional submissions reduces administrative burdens, allowing regulatory bodies to focus resources on evaluating novel devices and high-risk modifications.
Enhanced Device Performance: Continuous improvements through iterative updates can lead to more accurate diagnostics and treatments, ultimately improving patient outcomes.
Global Harmonization: The FDA, in collaboration with international regulatory agencies like Health Canada and the UK’s Medicines and Healthcare products Regulatory Agency (MHRA), has identified guiding principles for PCCPs. This collaborative approach promotes international harmonization, benefiting manufacturers operating in multiple markets.
Considerations for Manufacturers:
While the PCCP framework offers greater flexibility, manufacturers must ensure that:
Risk Management: Modifications do not introduce new risks or compromise device safety.
Transparency: Clear documentation and communication with regulatory bodies are maintained throughout the device’s lifecycle.
Compliance: All changes remain within the scope of the authorized PCCP, and any modifications beyond this scope require a new marketing submission.
Steps for AI Medical Device Manufacturers to Leverage the FDA’s Predetermined Change Control Plan (PCCP)
Manufacturers developing AI-enabled medical devices must follow a structured approach to incorporate the FDA’s Predetermined Change Control Plan (PCCP) into their regulatory strategy. Below is a step-by-step guide:
1. Identify AI-Driven Modifications
AI models evolve over time as they learn from new data. Manufacturers should categorize possible changes that their software may need to undergo, such as:
- Performance improvements: Refining accuracy and reducing false positives.
- Algorithm updates: Upgrading deep learning models based on new datasets.
- Expansion to new patient populations: Enhancing generalizability across different demographics.
- Changes in input data types: Allowing the AI to process new imaging modalities.
2. Develop a Comprehensive PCCP
The manufacturer should create a Predetermined Change Control Plan that includes:
- Description of the planned modifications (e.g., supporting a different CT scanner).
- Validation strategies to ensure that these modifications do not introduce risks.
- Performance metrics to evaluate the safety and effectiveness of updates.
- Protocols for monitoring and reporting AI performance after deployment.
3. Align with FDA’s Risk-Based Approach
The PCCP should focus on safety and effectiveness while ensuring that updates do not compromise patient outcomes. This means:
- Establishing pre-defined validation criteria for AI updates.
- Outlining risk mitigation strategies for potential algorithmic bias.
- Demonstrating compliance with Good Machine Learning Practices (GMLP).
4. Submit PCCP as Part of the Initial Marketing Application
Manufacturers should include the PCCP in their original premarket submission (510(k), De Novo, or PMA). This allows the FDA to assess whether the proposed modifications:
- Are well-defined and predictable.
- Have appropriate risk control measures.
- Do not significantly change the device’s intended use or performance.
5. Implement Post-Market Monitoring & Real-World Performance Analysis
Even after FDA clearance, manufacturers should continuously:
- Monitor AI model drift to detect unexpected performance issues.
- Validate AI predictions on new populations to prevent bias.
- Ensure ongoing compliance with regulatory frameworks.
Benefits of PCCP Adoption
To fully utilize the Predetermined Change Control Plan (PCCP), AI medical device manufacturers must ensure that any modifications remain within the device’s originally approved intended use. This means that expanding applicability to new organs, new diseases, or significantly altering functionality would require a new submission rather than relying on a PCCP.
With that in mind, here are examples of modifications that would (and would not) be permissible under a PCCP while still aligning with FDA regulations: