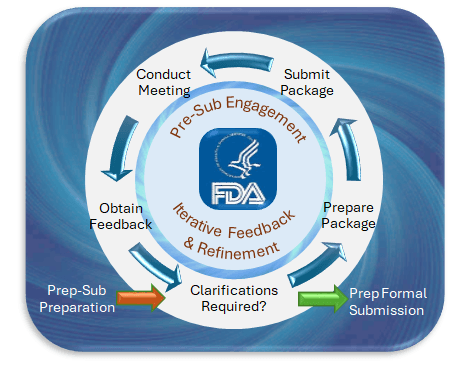
Agile Regulatory Engagement
The Benefits and Considerations of FDA Pre-Submission Meetings Navigating the regulatory landscape for medical devices can be complex and challenging. One invaluable tool to streamline this process is the FDA’s Pre-Submission (Pre-Sub) program. This program offers medical device manufacturers an opportunity to engage in early and constructive dialogue with the FDA, potentially smoothing the path…